|
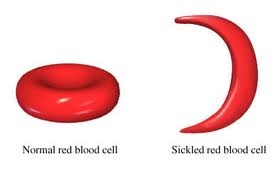
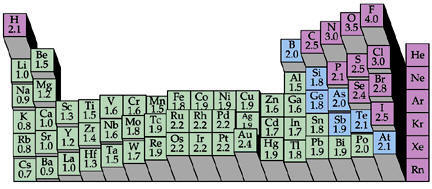
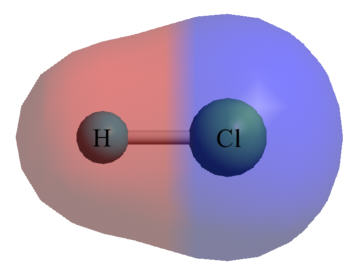
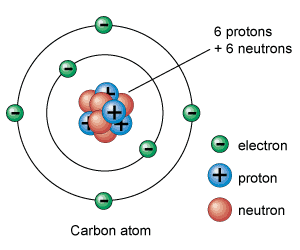
One of Pauling’s key discoveries in the field of chemistry was electronegativity: the degree to which each element attracts the electrons in a covalent bond. Atoms are composed of three types of subatomic particles: protons, neutrons, and electrons. Protons, which are positively-charged, and neutrons, which have no charge, are clustered together in the atom’s nucleus. In contrast, minuscule, negatively-charged electrons orbit the nucleus in patterns known as orbitals. A covalent bond forms when two atoms share electrons. When this occurs, the electrons are simultaneously attracted to the positively-charged nuclei of each of the atoms, and thus hold the nuclei together. For years, chemists had believed that the electrons in a covalent bond were shared equally between the two atoms. However, in 1932, Pauling published a paper outlining his theory that electrons were often shared unequally; that is, that one of the two nuclei would often attract them more than the other would. Pauling termed this attraction electronegativity, and developed a scale to illustrate the relative degree of attraction each element held for shared electrons. Hydrogen was assigned an electronegativity of 2.2, and values for the other elements were determined based on their ability to pull electrons away from hydrogen. The most electronegative element is fluorine, with a value of 4.0 on the Pauling scale, while the least is francium, with a value of 0.7. Therefore, in a bond between fluorine and hydrogen, electrons will spend the bulk of their time near fluorine, while they will concentrate near hydrogen in a francium-hydrogen bond. The introduction of Pauling’s theory of electronegativity also led to the study of polarity in bonds and molecules, another landmark that fundamentally altered the way chemists viewed molecular bonds. With the discovery of electronegativity, chemists realized that bonds, except those between atoms with identical electronegativities, were inherently unequal, with one particle attracting electrons more than the other. Therefore, even in a covalent bond, one of the atoms possessed a partially negative charge, while the other was partially positive, a condition known as bond polarity. For example, the fluorine in a hydrogen-fluorine bond would have a slightly negative charge because of its greater attraction of the electrons, leaving the hydrogen slightly positive. Yet, while electronegativity is Pauling’s best-known discovery among students of chemistry, the theory of orbital hybridization may have been his most significant contribution to the field. The carbon atom has six electrons, two of which orbit near the nucleus within a tight sphere, two of which orbit in a slightly larger sphere, and two of which move by themselves within individual orbitals. Since single covalent bonds form when each atom donates one electron, carbon should only have been able to bond twice, once with each of its individually-orbiting electrons. However, during Pauling’s era, experiments had shown that carbon could, in fact, bond four times. To reconcile this contradiction, Pauling theorized that, when a carbon atom bonds, its orbitals are altered so that the atom possesses four individually-orbiting electrons. Termed orbital hybridization, this theory was applied to all other elements, and was overwhelmingly supported by countless studies during the following decades, proving critical to further research into atomic and molecular structures. Pauling discovered that the cause of Sickle-Cell Anemia was a problem with the structure of the Hemoglobin protein in the blood cell.
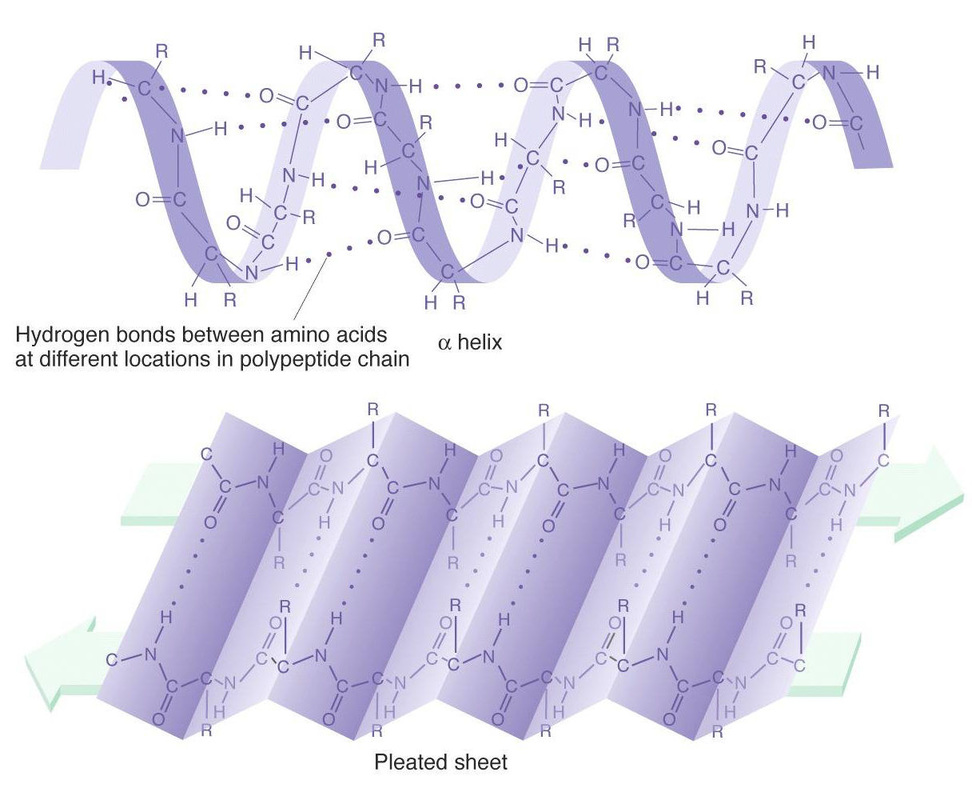
A diagram showing the Alpha Helix and the Beta pleated sheet.
|
A periodic table that shows electronegativity values. The taller the column, the higher the element's electronegativity.
An image of the electronegativity of a hydrogen-chlorine bond. The electron cloud surrounding the chlorine atom is larger because chlorine has a stronger attraction to electrons than hydrogen. The Cl end of the molecule is more negative because it has more of the electrons.
A diagram of a Carbon molecule, with its electrons
In the wake of his achievements in the field of bonding, which would make Pauling’s reputation as the world’s greatest living chemist, Pauling moved toward the advancing field of biology. With his application of chemical principles to biological systems, Pauling, along with others, founded the field of molecular biology, which provided the basis for modern studies of genetics and cancer. In one of his first forays into biology, Pauling outlined the allosteric nature of the hemoglobin molecule, which transports oxygen throughout the body and is therefore critical to survival. Pauling discovered that when an oxygen molecule binds to hemoglobin, the latter responds by changing shape to become more receptive to oxygen, which is known as an allosteric response. In contrast, when the pH, or acidity, of the blood increases, hemoglobin loses its affinity for oxygen and deposits it in cells. This is crucial to gas exchange because carbon dioxide changes into carbonic acid as it leaves cells and enters the bloodstream, on its way to the lungs and out of the body. As carbonic acid buildup increases the pH of the blood, hemoglobin deposits oxygen into our cells, balancing the body’s gases.
From this initial discovery, Pauling moved into more ambitious investigations, specifically of protein structure. Scientific legend has it that Pauling uncovered one of the two key underlying amino acid configurations in proteins, the alpha helix, by folding a sheet of paper while sick in bed. Proteins, the most complex biological molecules in our bodies, are composed of repeating but non-identical units called amino acids. The sequence of amino acids in a chain is referred to as a protein’s primary structure, which determines the pattern of the protein’s folds. Two common folding patterns, the alpha helix and beta pleated sheet, are termed the secondary structure of proteins, and were both discovered by Pauling and colleague Robert Corey. Amino acid chains fold into alpha helices when every fourth amino acid binds to another with hydrogen bonds, weak intermolecular attractions. This gives the chain a three dimensional helical structure, much like a single strand of DNA. In contrast, a different bonding pattern causes the beta pleated sheet to resemble a piece of paper folded “accordion-style,” forming a generally flatter chain. With models of the alpha helix and beta pleated sheet in hand, chemists, including Pauling, were able to pursue the structures of entire proteins over the following decades, leading to major advances in molecular and cell biology. |